Why LIMS is Not a Long-Term Archive
In life sciences environments, Laboratory Information Management Systems (LIMS) are essential. They manage samples, orchestrate workflows, and capture critical laboratory data. But while LIMS is indispensable for day-to-day operations, it was never designed to serve as a long-term archival platform.
That distinction matters particularly in the context of regulatory expectations for data integrity and retention.
Operational Control is Not Long-Term Preservation
A LIMS is fundamentally an operational system. It is built to enable change: new studies, new configurations, system upgrades, instrument integrations, workflow optimisation. Over time, it evolves. Databases are restructured, versions are updated, vendors are acquired, and infrastructure is modernised.
Long-term archiving and preservation on the other hand is about ensuring that data generated today remains complete, accessible, readable, and trustworthy decades into the future often long after the original system has changed or been retired.
These are different design objectives.
Regulators consistently emphasise that computerised systems must be “fit for purpose.” Validation alone does not guarantee long-term suitability. A system that has been validated to manage laboratory workflows may perform its intended operational functions correctly, yet still be inadequate for preserving secure, immutable, and regulator-ready records over 10, 20, or 30+ years. Validation confirms that a system works as specified; fitness for purpose confirms that it remains suitable for its intended use, including long-term data retention and integrity.
Regulatory Expectations: Data Must Endure
In regulated laboratories, some LIMS data must be kept long after day-to-day work has finished. This includes sample and batch records, test results, QC data, method versions, validation data, and audit trails.
- Under 21 CFR Part 211 (US GMP), batch and laboratory records must be kept for at least one year after the product’s expiry date.
- Under 21 CFR Part 58 (US GLP), study records must be retained for five years.
- Under Regulation (EU) No 536/2014, clinical trial documentation must be kept for at least 25 years.
- EU GMP guidance under EudraLex Volume 4 also ties record retention to product expiry and lifecycle.
Electronic records must also meet 21 CFR Part 11 requirements to ensure they remain secure and audit-ready throughout the retention period.
What “Post-Expiry” Means
“Post-expiry” means after the product’s labelled expiration date not after it was made.
If a product has a five-year shelf life and the rule says “one year post-expiry,” the data must be kept for at least six years from manufacture. If shelf life is extended, the retention period extends too.

+ 1 year post-expiry
Keep Data: 6 Years Total
What “Lifecycle of the Product” Means
Some records, especially validation and method history, must be kept for the full lifecycle of the product.
That lifecycle includes development, approval, manufacturing, time on the market, and even after the product is discontinued.
For long-lived products, this can mean 20–30 years or more.
When lifecycle rules and post-expiry rules are combined, organisations may need to keep LIMS data for decades. That is far longer than most operational systems are designed to last.

Keep Data: 20-30+ Years
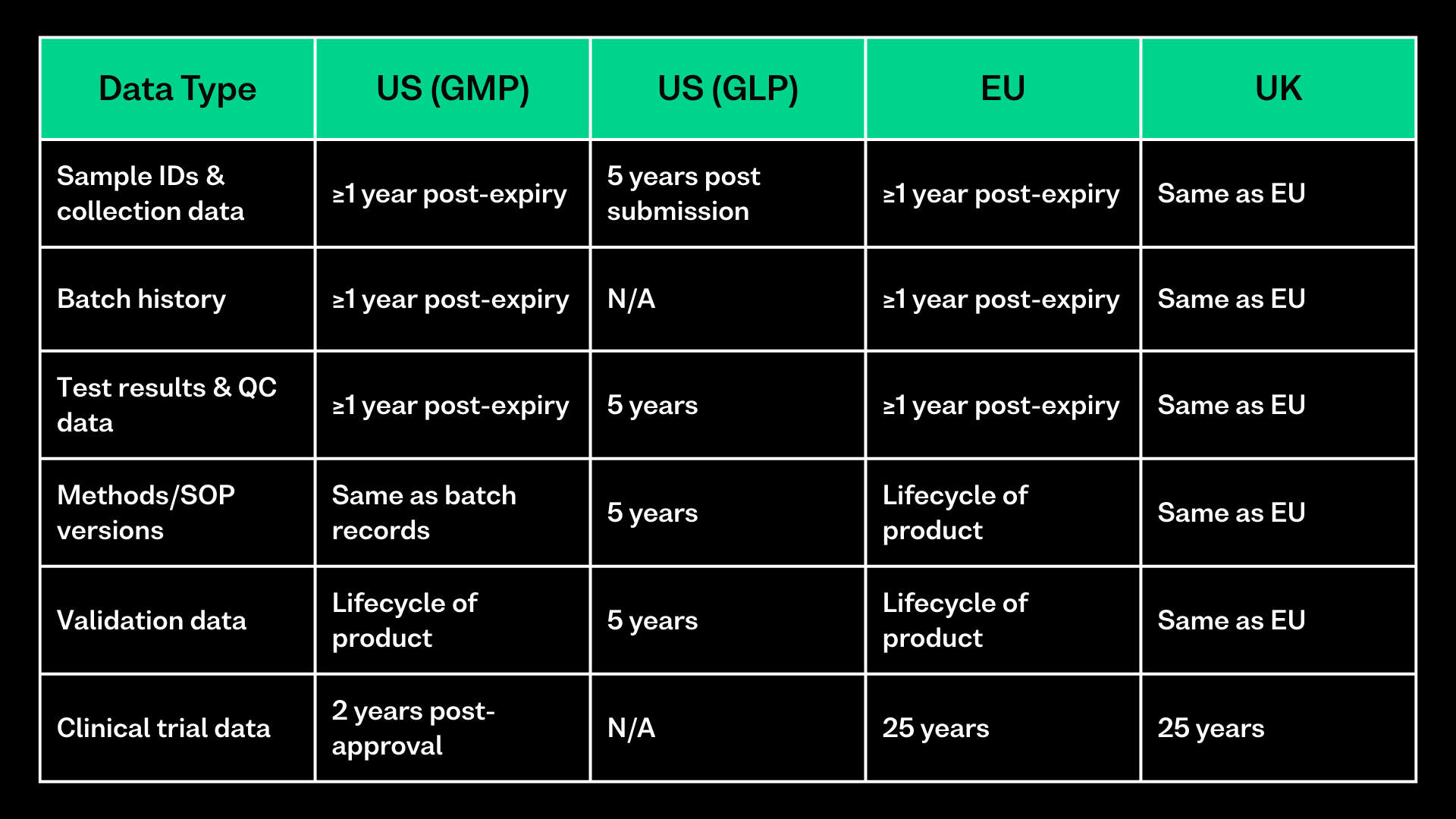
It is important to note that for internationally conducted studies, many organisations retain data for the longest applicable period. For example, while GLP regulations in the US may require validation data to be kept for 5 years, if the product’s lifecycle spans 20 years, organisations typically retain the data for the full 20 years to ensure compliance.
The Risk of Treating LIMS as the Archive
Using LIMS as a de facto archive introduces structural risk. Organisations may find themselves maintaining legacy systems purely to meet retention obligations. System upgrades can introduce complexity in maintaining legacy data structures. Decommissioning becomes costly and high risk, with significant effort required to extract complete, contextualised records in a regulator-ready form.
In inspections, the question is not whether data once existed it is whether it can be produced, in a complete and trustworthy form, when requested.
Summary
A LIMS is fit for purpose as an operational laboratory platform. It is not inherently fit for purpose as a long-term archive.
Separating operational systems from long-term preservation strategy reduces regulatory risk, controls lifecycle cost, and strengthens data integrity across the full data lifecycle.
With increasing regulatory scrutiny and growing data volumes, organisations must move beyond the assumption that “the system” is the archive.

Anthony Wells
Anthony assumed the role of Product Marketing Manager at Arkivum in 2024, leveraging over a decade of experience of product marketing management in the technology sector. Proficient in developing and executing marketing strategies, Anthony is also experienced in product lifecycle management, from inception through to discontinuation.
Get in touch
Interested in finding out more? Click the link below to arrange a time with one of our experienced team members.
Book a demo