Research Report: Transforming the Future of Clinical Data
Arkivum publishes new 2022 research report into clinical trial data.
 It’s now the third year that we have run our annual survey exploring the key trends within clinical data management. Building upon the foundations of the first two iterations, this year we broadened the scope from a specific focus on the TMF to look at the wider clinical trial data space.
It’s now the third year that we have run our annual survey exploring the key trends within clinical data management. Building upon the foundations of the first two iterations, this year we broadened the scope from a specific focus on the TMF to look at the wider clinical trial data space.
Following the digital transformation necessitated by the COVID-19 pandemic, we have seen a shift in priorities to data strategies over the past two years. However, many challenges and misconceptions still remain and the 2022 Transforming the Future of Clinical Data report brings some of these issues to light.
Some of the key findings from this year’s report includes:
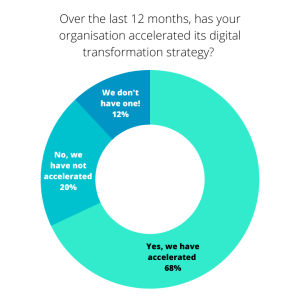
68% Life Sciences organisations say they have accelerated their digital transformation strategies by adopting new technology to improve efficiencies, compared to just 58% in our 2021 report. Following on from this speed of change, we asked respondents about their main transformation priorities over the next 12 months.
The top three responses were:
- 52% said ‘To streamline processes and improve productivity.
- 46% said ‘To reduce costs’ – up from just 29% in 2021.
- 34% said ‘To improve the management and conduct of trails’ – the same as last year.
Looking at these results there is a clear message that organisations within the sector are hoping that these new approaches and technologies will increase efficiency within the trial, while bringing overall costs down.
You can read the full guide for free by clicking here.

Tom Lynam
Tom is the Marketing Director at Arkivum. He joined the business in January 2020 tasked with driving new business growth and building the brand into new sectors such as Pharmaceutical and Life Sciences. He has over 12 years’ experience in several diverse marketing leadership roles across technology and professional services organisations.
Get in touch
Interested in finding out more? Click the link below to arrange a time with one of our experienced team members.
Book a demo